Thus, careful control of the reaction conditions allows exploitation of both pathways for the synthesis of different classes of heterocyclic derivatives. In addition an unprecedented reactivity of DCP as a very reactive heterocyclic electrophile at the C-4 carbon was also evidenced: by changing the experimental conditions, cyanopyrrolyl- and cyanoindolyl-pyridazines were obtained through reactions of pyrrole and indole systems as carbon nucleophiles in formal SNAr2 processes where a CN group of DCP acts as leaving group. AM1 and B3LYP calculations were performed for the DielsAlder reactions of a series of d-glucose-derived dienophiles with cyclopentadiene. HDA reactions with heterocyclic dienophiles allowed direct benzoannelation: in particular, pyrrole and indole derivatives were converted to dicyano-indoles and -carbazoles. 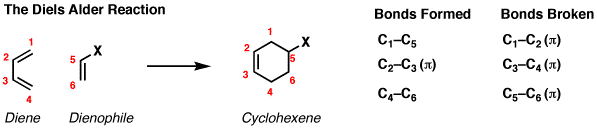
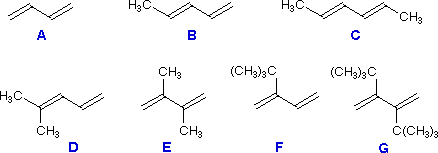
In this video, reactivity of different dienes and dienophiles has been discussed. 
The rate is affected by the nature of diene and dienophile. These findings again demonstrate the generality of the Pauli repulsion-lowering catalysis concept. The use of alkenes, alkynes and enamines as 2p electron counterparts afforded dicyanocyclohexa-1,3-dienes and substituted phthalonitriles, respectively, while the use of suitable bis-dienophiles provides a general strategy for the one-pot synthesis of polycyclic carbo- and hetero-cage systemsthrough pericyclic three-step homodomino processes. There are two components in DA, Diene and dienophile. Notably, the reactivity can be further enhanced on going from a Period 3 to a Period 5 LA, as these species amplify the asynchronicity of the Diels-Alder reaction due to a stronger binding to the dienophile. Despite the poor reputation of electron-deficient pyridazines in intermolecular Hetero Diels-Alder (HDA) reactions, 4,5-dicyanopyridazine (DCP) showed a surprising reactivity as a heterocyclic azadiene in inverse electron-demand HDA processes with different dienophiles.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
